UK medicines regulator investigates peptide clinics
Guardian finds unlicensed injections marketed with medicinal claims, websites edited after scrutiny but subscription model persists
Images
 There has been a boom in interest in using peptides for therapeutic purposes, from weight loss to anti-ageing and injury recovery. Photograph: Iuliia Burmistrova/Getty Images
theguardian.com
There has been a boom in interest in using peptides for therapeutic purposes, from weight loss to anti-ageing and injury recovery. Photograph: Iuliia Burmistrova/Getty Images
theguardian.com
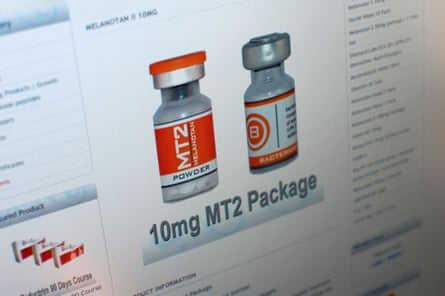 An ad for a peptide product that claims to help with skin tanning. Photograph: Richard Watkins/Alamy
theguardian.com
An ad for a peptide product that claims to help with skin tanning. Photograph: Richard Watkins/Alamy
theguardian.com
 Tirzepatide is found in the weight-loss drug Mounjaro. Photograph: Indranil Mukherjee/AFP/Getty Images
theguardian.com
Tirzepatide is found in the weight-loss drug Mounjaro. Photograph: Indranil Mukherjee/AFP/Getty Images
theguardian.com
The UK’s medicines regulator has opened inquiries into private “peptide clinics” after a Guardian investigation found providers advertising injectable peptides with claims ranging from “cognitive enhancement” to faster injury recovery. The Medicines and Healthcare products Regulatory Agency (MHRA) said that when clinics make medicinal claims, the products can be treated as medicines and fall under the Human Medicines Regulations 2012—triggering enforcement.
According to the Guardian, interest in peptides has surged in recent years, pushed by wellness marketing, social media influencers and some clinicians. The gap between demand and evidence is wide: much of the research cited in marketing is pre-clinical—cell or animal studies—while robust human trials are limited for many of the compounds being sold. Yet the market is packaged as a consumer service: clinics offer “consultations”, list named peptides, quote prices and describe expected “results duration”, while simultaneously labelling products “research only”.
That split language is a business strategy as much as a scientific caveat. If a clinic can sell a monthly subscription—£350 for one peptide or £450 for two, as one provider told the Guardian—while avoiding the regulatory obligations that come with licensed medicines, it can move faster than the evidence base and externalise the downside risk. The MHRA’s position is simple: you cannot advertise an unregulated injectable as if it treats, prevents or improves a medical condition without stepping into medicines law. The Guardian reports that after being contacted for comment, at least one clinic removed specific claims from its website—suggesting the marketing is sensitive to scrutiny.
The clinics’ own disclosures also show the limits of the product. In a consultation described by the Guardian, a clinician told a reporter that peptides often lack randomised, multi-centre trials and that long-term effects are uncertain. The same clinician nonetheless recommended combining peptides for post-exercise recovery and fatigue, and suggested cycling on and off them to “reduce any risks”. That is a familiar pattern in loosely regulated health markets: risk is acknowledged in general terms, while the sales pitch remains specific.
For regulators, the hard part is not defining the rule but policing a fragmented sector. Search-driven advertising means a clinic can become a top result overnight; claims can be edited quickly; and “wellness” framing allows medical-sounding promises to be presented as lifestyle optimisation. Enforcement tends to be reactive and case-by-case, while the commercial incentive is to test the boundary until someone intervenes.
The MHRA said it will take action where clinics are identified as breaching legal requirements. The Guardian’s reporting suggests at least some clinics have already started rewriting their websites—without changing the underlying business model of selling unlicensed injectable therapies on a recurring fee.
In the Guardian’s account, one clinic warned a prospective client about the lack of long-term data and then offered two peptides anyway. The price list remained.
