Quantum computer helps model half Möbius ring molecule
IBM-backed chemistry result mixes lab synthesis with hybrid computation, practical advantage remains narrow and benchmark-like
Images
 arstechnica.com
arstechnica.com
 Photo of John Timmer
arstechnica.com
Photo of John Timmer
arstechnica.com
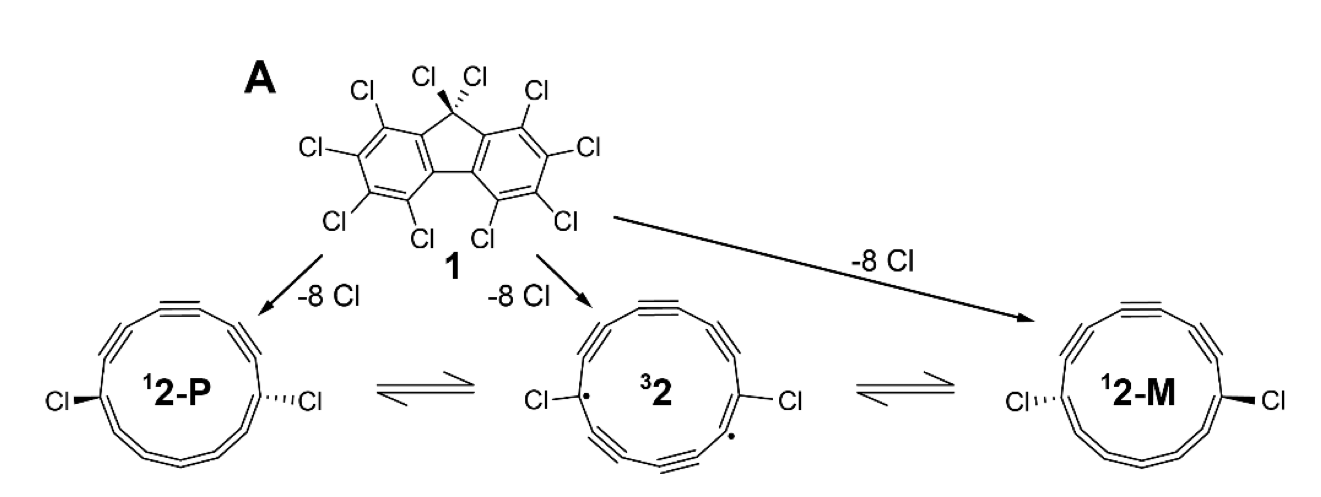
Quantum chemistry met a Möbius twist last week when IBM highlighted work on a “half‑Möbius” ring molecule whose electron orbitals do not return to their starting orientation after one lap. According to Ars Technica, the international team built a 13‑carbon ring with substituents arranged so an electron would need multiple circuits around the ring to come back to its original state—an exotic topology more familiar from a Möbius strip than from textbook benzene.
The molecule is not presented as a useful material; it is described as unstable and prone to collapsing into a more ordinary configuration. The practical point sits elsewhere: predicting the electronic structure of such a system is a stress test for today’s quantum‑chemistry methods because many interacting electrons must be treated together, not as nearly independent particles. Classical approaches can approximate these correlations, but accuracy tends to degrade as molecules grow and as the relevant orbitals become more entangled.
That is where the quantum‑computing angle enters. The paper used a quantum algorithm for part of the electronic‑structure calculation, alongside conventional computation and decades of chemical know‑how about how to steer orbitals with substituents. The computation did not “discover” the molecule on its own; it contributed to evaluating competing electronic configurations in a design space that becomes expensive to search with brute‑force classical methods.
Ars Technica frames the result as a snapshot of where quantum computers actually are: able to assist with narrowly scoped chemistry tasks, but not yet delivering a broad, routine advantage over well‑tuned classical software. Noise, limited qubit counts, and the need for careful approximations still dominate what can be run. Even when a quantum routine is used, the surrounding workflow—problem setup, choice of ansatz, error mitigation, and classical post‑processing—determines whether the output is chemically meaningful.
The Möbius molecule therefore functions less as a product demonstration than as a benchmark. It combines a physically demanding electronic structure with an experimental target that can be made—briefly—allowing theory, algorithm, and synthesis to be checked against one another. That kind of loop is what “useful quantum chemistry” will ultimately require: not a single spectacular calculation, but repeated predictions that survive contact with a lab bench.
In the end, the most durable fact in the paper is not the twisted orbital picture but the division of labor it implies. A fragile ring of 13 carbons can be made to behave like a topological puzzle, and a quantum processor can be made to carry a small part of the math, but the workflow still depends on chemists deciding what to build and classical computers doing most of the work.
